
The characteristics of the Nanoplant Implant:
The Nanoplant Implant System can be applied in the case of harder than average and thin bone structures for removable and glued toothworks as well.
Distinctive constructional and applicational characteristics: • Minimal invasive intervention- transgingival surgical technique • OT-CAP micro compatible snap head • Hybrid abutment system • Screw selectional possibility adjusted to the height of the sulcus • Geometry adjusted to the natural tooth root • Self-compressional, self- tapping and self-closing conical thread form •The cycloid cord thread at one-third of the screw exceptionally resists the dynamic power impact, gradually deepening and sharpening • High primer stability- immediate loadness, above 35 Ncm, high primer stability- immediate loadness, above 35 Ncm • Bauer type compatibility
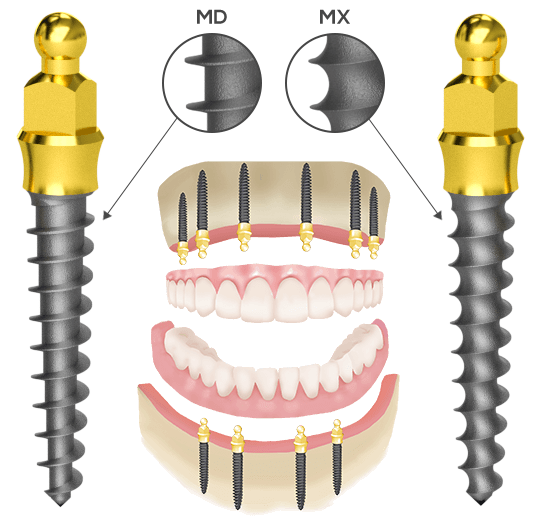
The Nanoplant Implant System consists of two types of single-phase implants:
• MX- For softer maxila bone structure with higher thread pitch
• MD- For harder mandibula bone structure with thin thread edge

The abutments of the Nanoplant System:
OT-CAP micro metal cap; OT-CAP micro plastic interline; Aesthetics (anatomical) head- straight; Aesthetics (anatomical) head -15 degree oblique
Applied raw materials
BIONIKA demonstrates the best qualities of Grade 4 titanium used in implant manufacturing for dental implantology according to the ISO 5832-2 ASTM F67 standard.
Due to its adequate purity the biocompatibility is exceptionally good as well as it is provided with exceptional solidity. Initially, we and other implant manufacturers preferred the higher purity titanium but due to solidity reasons nowadays almost every implant is made of Grade 4 or other alloyed titanium in the world.
In all cases of implant abutments, alloyed, high strength Grade 5 titanium is applied according to the ISO 5832-4 ASTM F136 standard. The quality of the products is guaranteed by design, manufacturing and quality management according to the harmonized European Union laws. The BIONIKA Medline Orvostechinkai Kft. is operated according to the EN ISO 9001 and the EN ISO 13485 quality management system. Our products are provided with CE marks, which is certified by the TÜV Rheinland.
The titanium used according to the standard is provided with exceptional biocompatibility, this is why it is almost risk-free.

